Thinking about metal Binder Jetting or FFF? Here is (almost) everything you need to know about sintering
With the arrival of high-volume metal Binder Jet systems and a growing interest in metal Fused Filament Fabrication, the AM industry is set for a new phase of growth. The ability to use this new generation of systems for the production of ‘green’ parts is, however, only half of the story. The sintering of these parts to create large quantities of finished product to a consistent quality requires both an investment in furnaces that can cost in excess of $1 million each, and a thorough understanding of sintering. In this article, Prof Randall German, the leading authority on the science of sintering, outlines the process and its core challenges. [First published in Metal AM Vol. 5 No. 3, Autumn 2019 | 35 minute read | View on Issuu | Download PDF]

Sintering is a heating process applied to shaped powders to provide strength by bonding the particles into a solid. Often, but not always, densification occurs during sintering. Surface diffusion forms the first sinter bonds without causing a dimensional change. When heated to a higher temperature, grain boundary diffusion induces densification, grain growth, pore rounding, and significant property changes. The optimal sintering conditions depend on the material and particle size, realising excessive heating leads to a loss of properties and even swelling of the component. Beyond classic textbook materials, sintered materials often rely on intentional grain boundary segregants, including liquids, to retard grain growth or accelerate sintering.
Within the confines of Additive Manufacturing, it is important to differentiate between the sintering process that is used to densify parts created by metal Binder Jetting and Fused Filament Fabrication, and the Laser Powder Bed Fusion of metal powders, often erroneously referred to as a variant of ‘laser sintering’, in which a very different melting process occurs.
The term ‘sintering’ arose from geology where ‘cinder’ was used to describe hardening of mineral phases around geothermal vents. Modern uses for sintering trace to the early 1800s and the fabrication of platinum crucibles for melting glass. By the early 1900s, sintering was used to fabricate tungsten ingots from which lamp filaments were drawn. Scientific understanding of how sintering occurred emerged as atomic diffusion became accepted in the late 1940s. Predictive models for the role of time, temperature, particle size, and green density emerged in a body of mathematical expressions by the 1980s [1,2]. Unfortunately, the models assume ideal conditions and often ignore significant parameters such as heating rate and atmosphere reactions. With modern instrumentation, it is clear that most sintering occurs during heating, not during the hold at the peak temperature.
The study of sintering spans a broad range of materials. In spite of its broad use, the sintering trajectory is essentially the same for most materials (polymers, ceramics, metals, composites, minerals and intermetallics). Thus, in teaching sintering behaviour, a broad range of examples and materials is available to help understand the general behaviour.
Industrial sintering cycles are defined by several parameters – green density, particle size, heating rate, peak temperature, hold time and process atmosphere. Pressure is applied in some cases, but pressureless sintering is predominantly favoured because of its significantly lower cost. Further variations arise in the design and operation of the sintering furnace. The peak temperature depends on factors such as the material melting temperature and particle size. The hold time at the peak temperature ranges from a few seconds to a few hours. Time at temperature can be adjusted to deliver equivalent thermal work; high temperatures require less time. Although the green body is weak prior to sintering, after firing it is strong, often competitive in properties with that attained via other manufacturing routes. Furthermore, the structure shows little evidence of the initial particles. Atmosphere selection is guided by the material chemistry and is a significant factor for proper sintering.
Binders are routinely added to powders to facilitate shaping. The binders are waxes or other common polymers that can be removed during heating to the sintering temperature – they decompose at temperatures far below the sintering temperature. Consequently, much of the sintering cycle is independent of the binder composition and component forming route, but it does depend on the powder chemistry and particle size. Thus, in spite of the availability of a diverse range of binders and shaping approaches, sintering cycles are often similar for seemingly different forming routes.
Formally, sintering is a thermal treatment applied to particles to increase strength by bonding particles to one another. The bonding occurs by atomic motion with a net reduction in surface energy as evident by surface area loss. It is possible to induce densification along with bonding, but a change in density is not always observed, especially for larger particle sizes. Filters and bearings are examples of sintered products where densification is intentionally avoided. Sintering is enhanced by smaller particles, higher temperatures and certain grain boundary phases. Although liquid formation is common during sintering, practically it is limited to less than about 15 vol.% liquid. Larger amounts correspond to semi-solid casting approaches such as rheocasting. Total particle melting with electron beams, lasers, or arcs is not sintering, but a variant of casting.
The basics of sintering
Atomic motion during sintering results in a loss of surface area and surface energy. Temperature controls the process. During heating at low temperatures, bonding occurs at the contact points between particles, as pictured in Fig. 1. This image shows necks between spherical bronze particles during early sintering. As the necks grow, there is initially no densification. Later, with higher temperatures, smaller particles or longer times, the particles pull together to fuse into a dense structure that no longer shows evidence of the precursor particles. Indeed, coarsening during sintering results in grains several times larger than the initial particle size. Hundreds and even thousands of particles fuse to generate a single grain.
For loose powder, such as that used in sinter-based Additive Manufacturing, sintering follows a geometric progression consisting of four stages:
- Contact formation – Weak adhesive forces pull the particles together
- Neck growth – Initial bonding at particle contacts by surface diffusion
- Pore rounding – Growing necks merge to form tubular pores
- Pore closure – Final stage grain growth with closed pores
After initial bonding, the pulling together at particle contacts, due to capillary stress, results in shrinkage and densification. In turn, shrinkage induces formation of new particle contacts that further bond the structure. For sinter-based Additive Manufacturing, the initial structure corresponds to each particle having five to six touching particles, termed the coordination number. As shrinkage occurs the coordination number increases to an average of about thirteen to fourteen faces on each grain at full density. Thus, sintered properties dramatically improve with densification, due to an increase in the interparticle bond size and number of interparticle bonds. The coordination number NC relates to the fractional density f as follows:
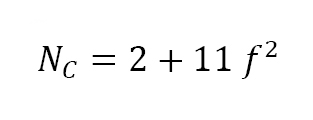
Density is the most common measure of the degree of sintering. It is a good predictor of properties since most materials follow a power law relation between strength σ and fractional density f:
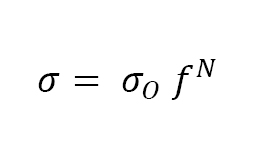
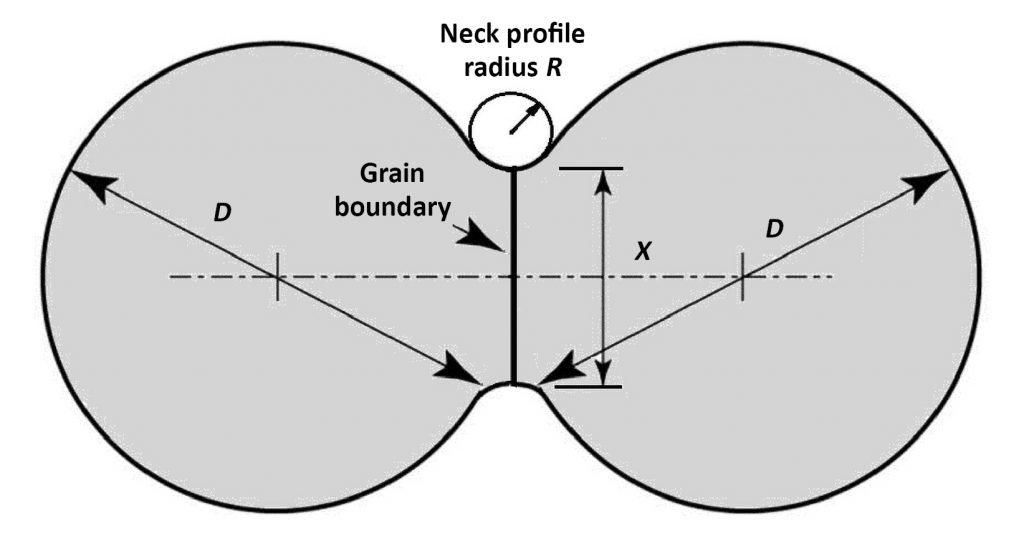
where σO is the strength at full density. The exponent N is often from 4 to 6, depending on the material sensitivity to residual pores. For ferrous alloys, this means 15% porosity (0.85 fractional density) lowers strength to 50% of the full density level (N ≈ 4.3). Besides density, other sintering monitors include shrinkage, conductivity and hardness. These parameters link to the neck size ratio; that is, the diameter of the sinter bond between particles X divided by the particle diameter D, giving the dimensionless neck size ratio X/D.
Shrinkage is the decrease in dimension ∆L from the green body size prior to sintering LO, giving ∆L/LO. It is often given as a percentage. Measures such as density and shrinkage are relatively easy to perform versus measuring neck size. A related parameter is densification, the change in porosity resulting from sintering normalised to the starting porosity. If all pore space is eliminated during sintering, then densification is 100%. These parameters are related and one can be used to calculate the others.
In most situations the sintered component volume is smaller than the green or initial volume, while mass remains essentially the same. The density increase associated with shrinkage (usually expressed as a positive value) is often assumed to be isotropic. If mass remains constant, the sintered fractional density f depends on the initial unsintered fractional density fO and shrinkage ΔL/LO as,
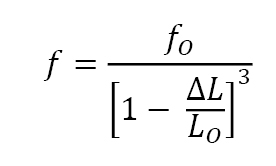
For a high green density, such as 85% of theoretical (possible using mixed particle sizes), a shrinkage of 3% gives 93% density and 5.3% shrinkage gives full density. On the other hand, at a green density of 45%, the required sintering shrinkage is 23.3% to reach full density. Generally the larger the shrinkage, the larger the distortion during sintering. A close look at sinter-based Additive Manufacturing approaches reveals density gradients in the green body, leading to anisotropic shrinkage. Other factors, including substrate friction and non-uniform heating, also contribute to sintered size variations.
Molecular dynamics computer simulations help visualise the atomic motion associated with neck growth. These simulations input atomic interaction potentials that govern atom motion during heating. Clusters of atoms are used to form particles. Each atom follows an interatomic attraction-repulsion potential that mimics melting, thermal expansion, and elastic properties. Atoms separated by more than the equilibrium spacing are attracted to one another, but atoms closer than the equilibrium spacing repel each other. Heat increases the atomic vibrational amplitude. During sintering the saturation vibrational frequency is near 1014 vibrations per second. As temperature increases, more atomic motion occurs, evidenced by increased vibrational amplitude. Initially, surface diffusion is active, where atoms move from convex surface sources to grow the concave neck. At the sintering temperature each atom changes position on average six times per second.
Sintering visualisation is possible as shown by the images in Fig. 2. These pictures correspond to tungsten particles heated to increasingly higher temperatures at a constant rate from room temperature to melting. As temperature increases the particles grow a neck, then shrink together. A grain boundary forms in the neck due to random misalignment of neighbouring crystals. These grain boundaries are critical to subsequent shrinkage and densification. Evaporation is evident at high temperatures. For two particles, the lowest energy configuration corresponds to coalescence into a single particle 1.26 times the initial size.
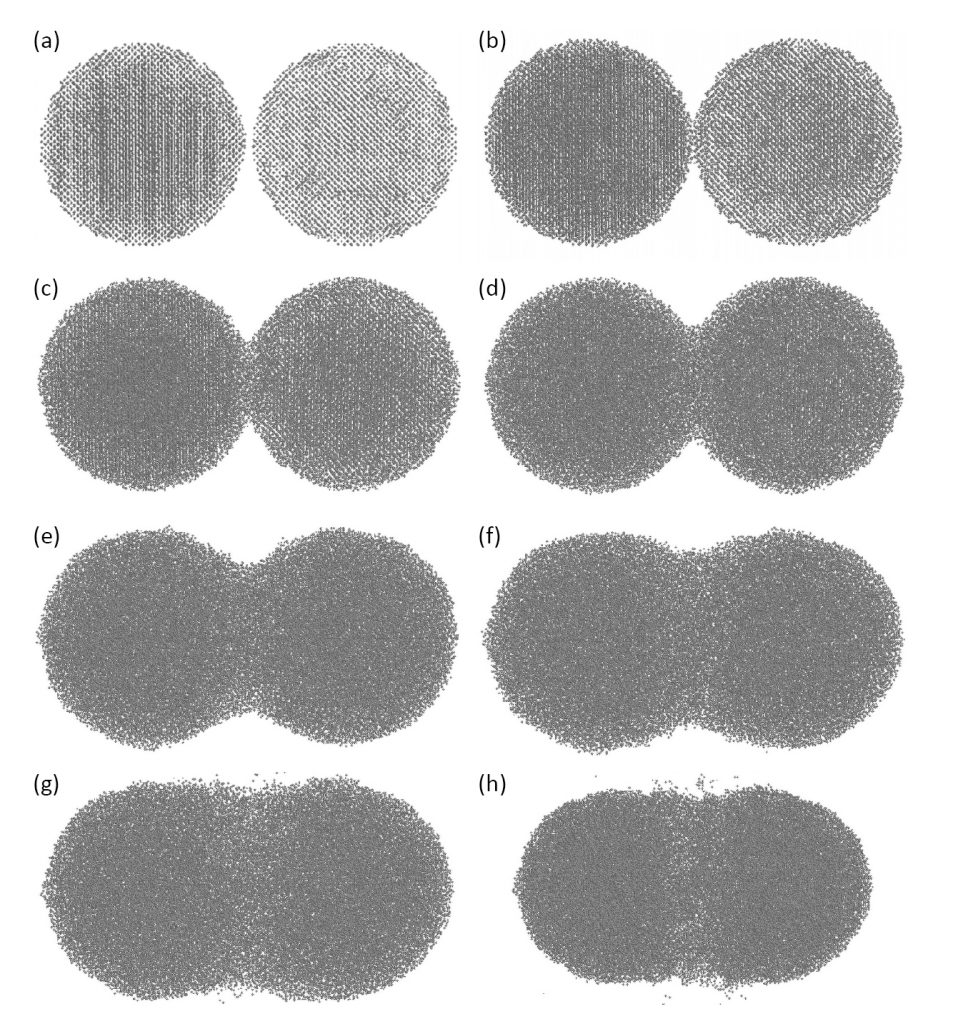
Such simulations show several aspects of sintering – atoms move, randomly, but that motion is biased over time by a progressive reduction in interface energy. At first this is by atom motion over the particle surface, termed surface diffusion. The overall action is to eliminate surface area by neck growth. As grain boundaries form between particles, grain boundary diffusion emerges. It is grain boundary diffusion that gives densification, since atoms move into the pores from the contact plane between particles, allowing progressive particle approach or shrinkage and formation of new contacts.
It is possible to accelerate sintering by adjusting the grain boundary chemistry. One common step is to form a liquid phase, since atomic motion is hundreds of times faster in liquids versus solids. Solid diffusion involves the creation of a vacant site for atomic diffusion and a similar energy to ‘jump’ an atom into the vacancy. In liquids, the amorphous atomic structure circumvents the vacancy creation energy. Thus, liquid phase sintering is easier, faster and widely employed in industrial sintering – by nature, humans are impatient.
Atomic motion
Sintering combines surface energy and high-temperature atomic motion to bond (surface diffusion) and densify (grain boundary diffusion) particles. What happens at the contact between particles is replicated billions of times throughout a component. Thus, models for bonding of two contacting particles give a glimpse at the behaviour, realising the events are replicated throughout a component.
The idealised two-particle sintering profile is shown in Fig. 3. The spherical particles are D in diameter with a neck X in diameter. Initially, the neck contour forms a concave saddle approximated by opposing radii of X and the circle of radius R. Surface energy reflects broken atomic bonds, so the smaller radius is higher in energy, generating a capillary stress that pulls the particles together to enlarge the neck. This capillary stress is termed the sintering stress. It is roughly proportional to twice the surface energy (usually 1–2 J/m2) divided by R. For a 10 µm powder, R is nominally about 1 µm so the sintering stress is about 2 MPa. That stress falls as the neck enlarges, but still it is sufficient to pull the contacting particles together up to about 95% density.
At room temperature, there is too little atomic motion to induce shrinkage or densification. However, at the sintering temperature there is considerable motion. For steel or stainless steel, sintering requires heating to a point where the material softens. This is termed thermal softening. Before significant bonding, a powder compact has a relatively low strength (in the 0.1 MPa range). Because of the low strength, fracture susceptibility is high during heating. Defects as small as 10 µm enlarge to become cracks; these form early prior to much necking. Differential shrinkage between thick and thin sections is a common difficulty, requiring slow heating rates to avoid cracks. Sinter bonding improves strength, but thermal softening at high temperatures reduces strength sufficiently to allow densification. That is, first the particles bond with no appreciable shrinkage (usually by surface diffusion), forming grain boundaries at the particle contacts. The neck growth increases the compact strength. Then, at higher temperatures grain boundary diffusion gives densification as the structure softens. Finally, remaining pores collapse into spherical voids that annihilate if they are not filled with an insoluble gas. This occurs late in the sintering cycle at high temperatures where the component is again weak due to thermal softening
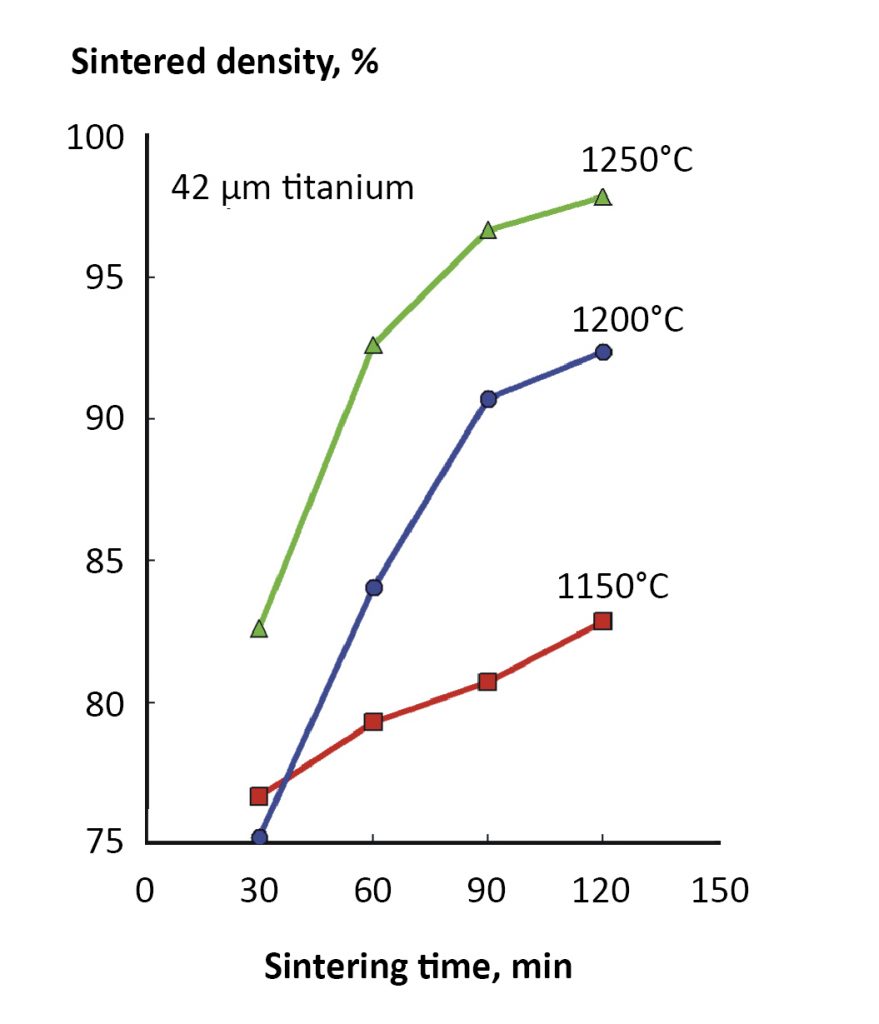
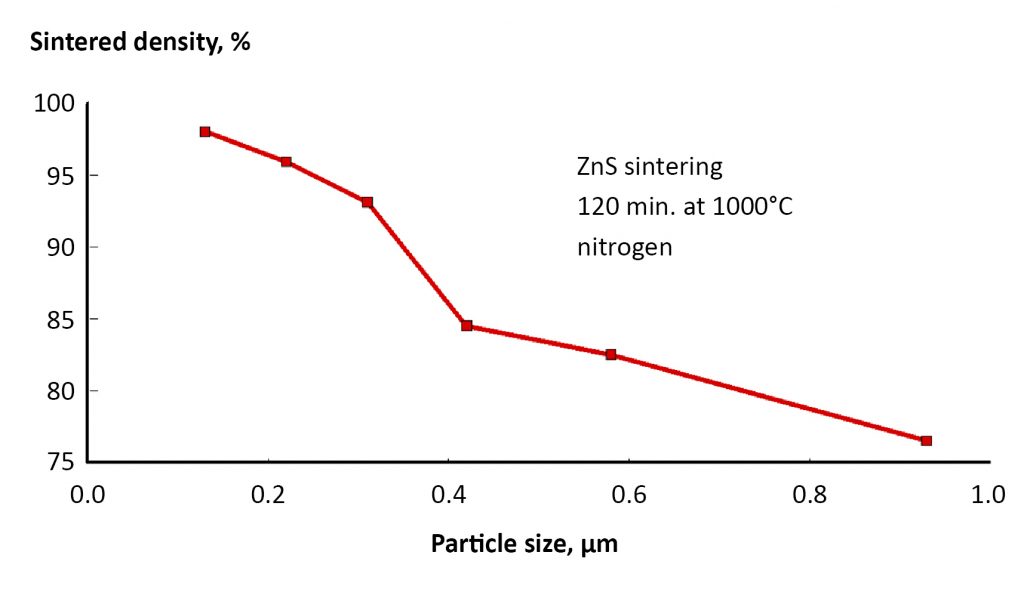
Sintering is faster at higher temperatures since atomic motion increases amplitude on heating. A practical demonstration is given in Fig. 4, where sintered density is plotted versus hold time for 42 µm titanium powder sintered at three temperatures [3]. Prolonged time adds to the densification, but the role of temperature is dominant. Smaller particles are helpful, since they have a higher sintering stress and shorter diffusion distances. This is demonstrated in Fig. 5 for experiments using different particle sizes of ZnS sintered at 1000°C for 120 min. in nitrogen [2]. Indeed, with a stainless steel powder it is possible to reach 98% density after 120 min. at 1050°C with a 4 µm powder [4]. But 1300°C is required with a 9 µm powder, and 1385°C for 31 µm powder. Smaller particles respond to a lower sintering temperature.
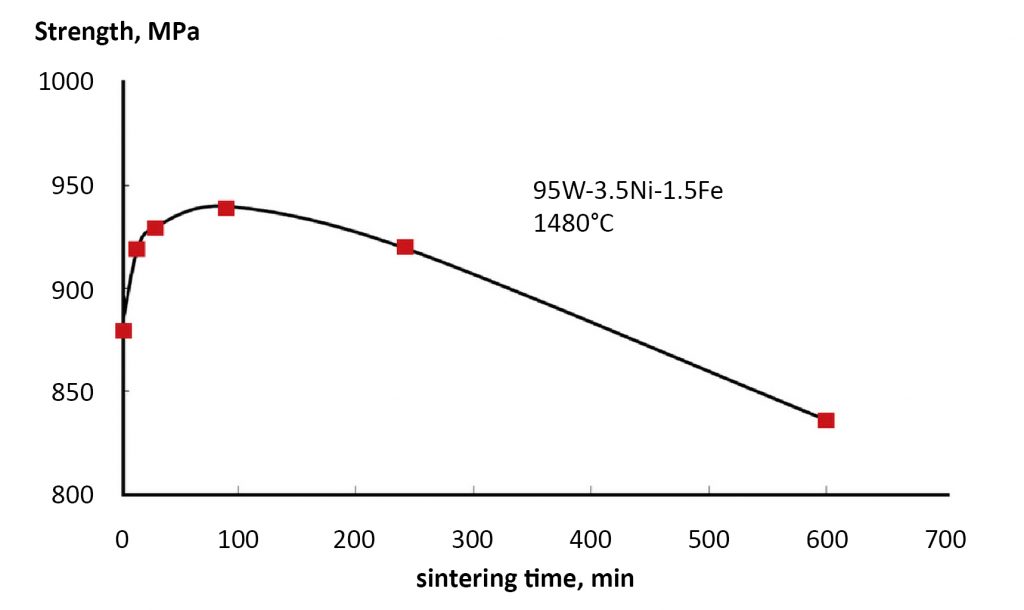
Due to grain growth during sintering, the final grain size is larger than the starting particle size. However, pore size is complicated. Porosity decreases due to densification, but simultaneously pores coarsen to increase size by vacancy or gas exchange, reducing the number of pores and pore surface area. With too high a sintering temperature, or too long a sintering time, the combination of pore coarsening and grain coarsening results in loss of density and properties, termed over-sintering. Fig. 6 plots such behaviour for a tungsten alloy sintered at 1480°C. Optimal strength is attained with less than a two-hour hold. Sintered stainless steel exhibit a similar loss of strength with excess sintering [4].
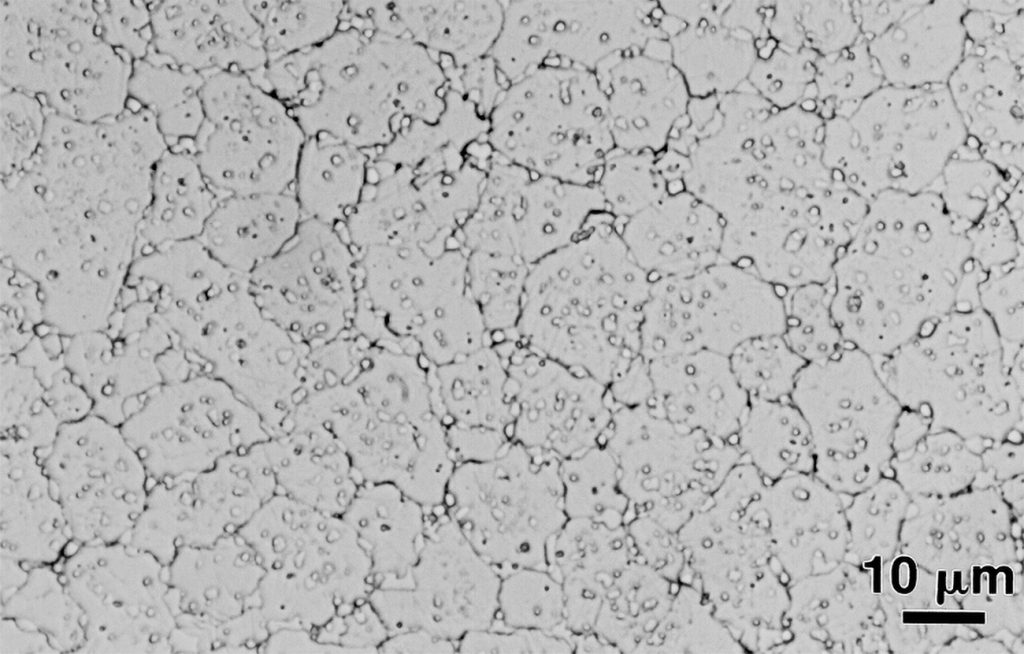
The driving energy for sintering is small, typically less than 1 J/g, resulting in a slow process. For some materials, such as WC-Co cemented carbides, this means heating to a temperature where liquid forms. Liquids that have solubility for the solid accelerate atomic motion and facilitate rapid sintering. Upon cooling, the liquid solidifies to give a composite microstructure consisting of solid grains bonded to one another with an interpenetrating second phase, as evident in the microstructure shown in Fig. 7. This stainless steel was doped with boron to form liquid on the grain boundaries during sintering. That liquid induced full density, without degrading properties. This is an alloy custom designed for sintering; boron reduces the sintering temperature by about 100 – 120°C. Table 1 shows a few examples of boron treatments [5-10]. Equivalent 12 µm powder without boron sintered at 1250°C gives a strength near 300 MPa and 18% elongation. The standard for sintered 316L stainless steel doped with boron is a strength of 520 MPa with 23% elongation [11]; for comparison, the industry standard for metal injection moulded 316L stainless steel is 450 MPa tensile strength and 40% elongation.

Liquid phase sintering relies on grain boundary wetting. Several examples are well known – TiC-tool steel, Fe-Fe3P, WC-Co, W-Ni-Fe, Mo-Ni, Fe-Cu-C, stainless-boron, Cu-Sn, and most ceramics. For example, AlN is treated with Y2O3 to form a fast diffusion grain boundary phase. Indeed, covalent ceramics such as SiC and Si3N4 only densify when additives are employed to form an amorphous or glassy liquid phase on the grain boundaries.
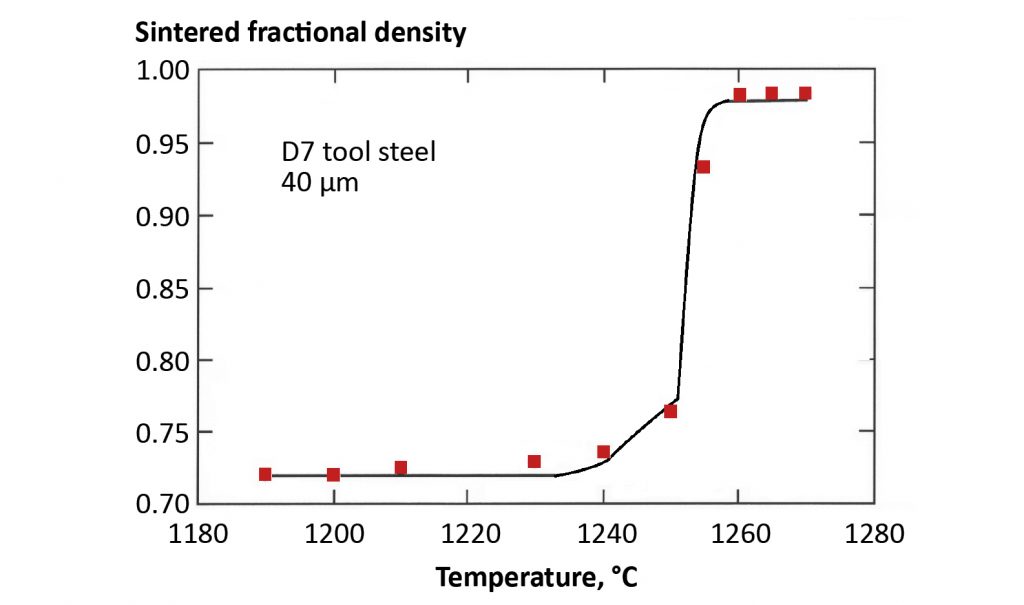
Alloy powders liquid phase sinter when heated to the solidus temperature where liquid forms inside the particles. This liquid softens the structure sufficiently that the sintering stress quickly pulls the particle to full density. Temperature controls liquid formation, as evident by the plot of density versus temperature in Fig. 8 for 40 µm D7 tool steel powder [12]. Densification occurs near 1250°C. Hold time is a minor factor. This supersolidus sintering approach is applied to high alloy systems, such as tool steels, stainless steels, cobalt-chromium, and superalloys; generally alloys based on iron, nickel, cobalt, precious metals, or titanium. Fig. 9 shows the microstructure for a 0.8% carbon M2 tool steel sintered at 1280°C in 10 min. using a nitrogen-hydrogen atmosphere. Supersolidus sintering requires precise temperature control because excess liquid causes component distortion.
There are two categories of atomic transport mechanisms – surface and bulk. Bulk transport along grain boundaries causes shrinkage, pore annihilation, and densification. Surface transport processes, mostly surface diffusion, reposition mass on the pore surface to smooth the pores and reduce surface area, but do not give densification. Such behaviour is useful in forming filters. In certain atmospheres (hydrogen doped with water or a halide such as chlorine), pore rounding without densification occurs by evaporation and condensation across pores. Otherwise, early sintering is predominantly by surface diffusion and later densification is by grain boundary transport. The exceptions are amorphous materials, such as glasses or plastics, that sinter by viscous flow.
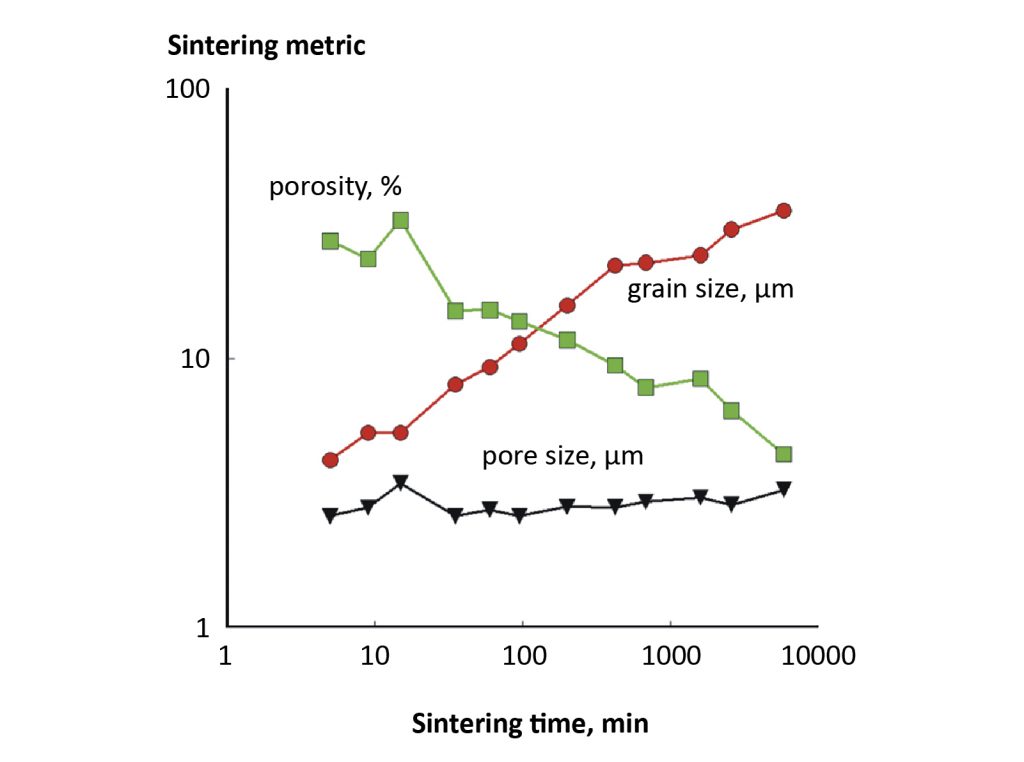
In sintering science, the pores are assumed to be accumulations of vacancies. Pores emit vacancies, just like a helium balloon gives up gas. These vacancies are annihilated at free surfaces and grain boundaries. Atoms move in the opposite direction to fill the pores. Vacancies also migrate between pores, leading to the growth of larger pores while the smaller pores shrink. Fig. 10 plots such behaviour using log-log scaling for 4.5 µm iron powder sintering at 850°C [13]. Porosity decreases, grains grow, and after an initial transient the grain boundary area decreases while the number of pores decreases. The pore size enlarges progressively even though densification reduces the total porosity and number of pores. As grain growth occurs, the grain boundary area declines, leading to slower sintering.
Control parameters
Of the several parameters that control sintering, temperature is dominant. Fig. 4 previously illustrated this effect. Shrinkage increases with higher temperatures, but the shrinkage rate slows with time. Such behaviour is treated using models where the monitor Y (such as shrinkage, neck size ratio, surface area loss, or densification) depends on hold time t and absolute temperature T via the relation:
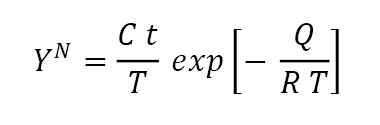
Here T is the absolute temperature, Q is an activation energy (proportional to the material’s melting temperature), R is the gas constant, and C is a material-geometry constant. An example of the shrinkage relation to time and temperature is given in Fig. 11, where the shrinkage data correspond to an exponent N = 3.
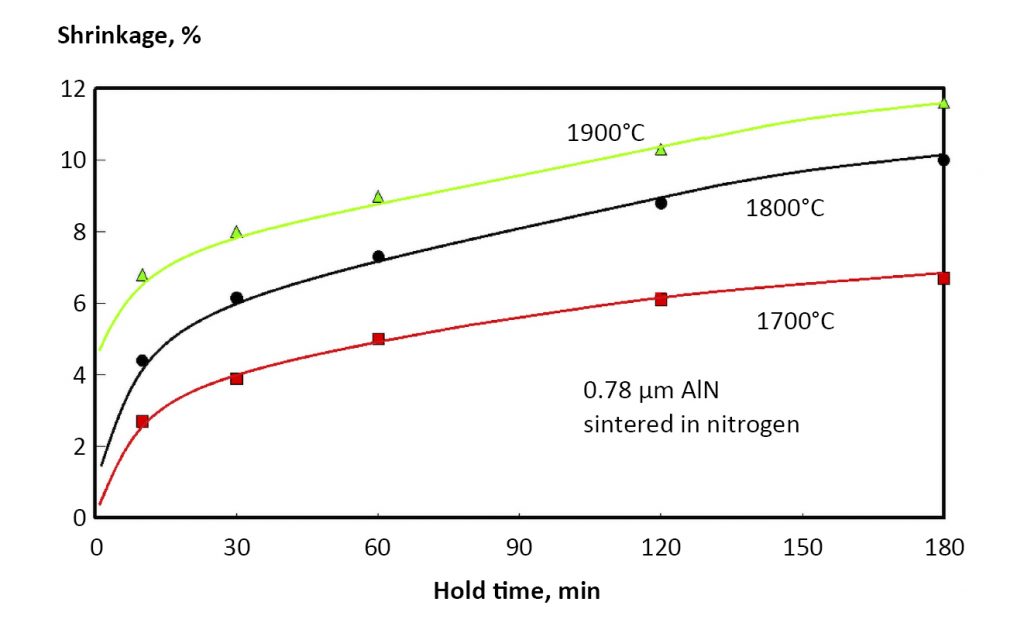
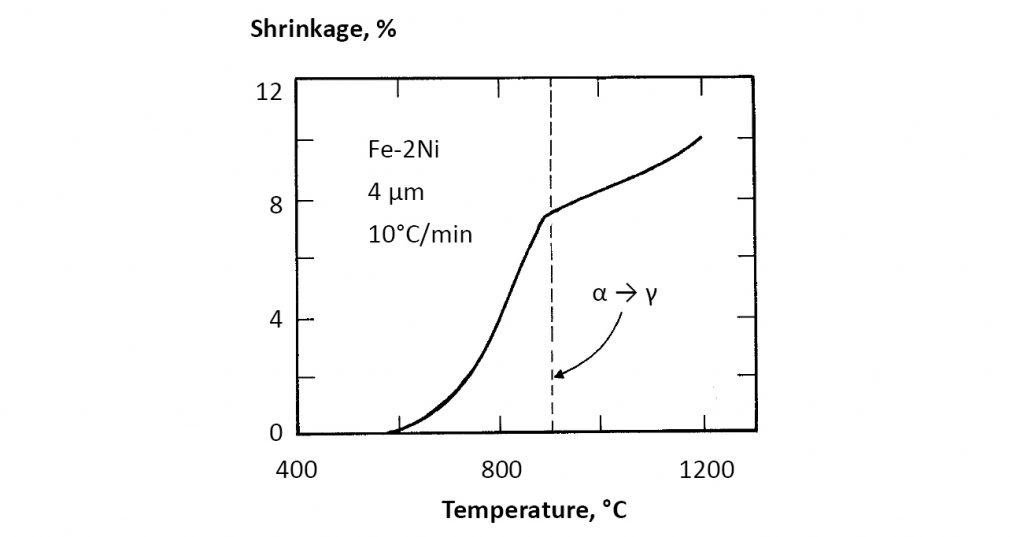
The factor C depends on the material chemistry and particle size. If a phase transformation occurs, such as iron from body-centred cubic to face-centered cubic, then the rate of sintering changes due to shifts in diffusion rates, grain size and formation of new interfaces. This effect is seen in Fig. 12 for Fe-2Ni heated at 10°C/min. through the body-centred cubic to face-centered cubic phase transformation (alpha to gamma). The data were taken using dilatometry, where dimensions are measured directly during sintering. In this case, most of the shrinkage occurs in the lower-temperature body-centred cubic phase, slowing after the phase change. However, steels require sintering in the high-temperature face-centred cubic domain in order to adsorb carbon for strengthening. Thus, for many ferrous alloys the typical sintering temperature is 1150 – 1350°C.
Hold time at the peak temperature is not a strong factor. Sufficient time is required to ensure uniform heating, but long times have diminished impact. Indeed, with respect to properties, long sintering times are counterproductive due to grain and pore growth. Some industrial sintering cycles are very short. For example, electronic films are sintered in a flash lasting only seconds. However, for bulk shapes, rapid heating induces thermal stresses that cause distortion. In metal injection moulded 316L stainless steel, the dimensional scatter increased with heating rate [14]. Thus, heating rates of 10°C/ min. or less are more typical.
Green density is another processing parameter. Smooth particles improve green density. A lower green density makes sintering more difficult since there are fewer particle contacts. It takes less net atomic motion to densify a well-packed, high-coordination number compact. Loose packing only reaches about six to seven contacts per particle. For spheres an idealised packing reaches a coordination number of twelve, resulting in improved sintering [15]. An example with 316L stainless steel powder contrasts sintered density versus initial packing (1360°C for 60 min. in hydrogen):
- 66.4 vol.% packing sinters to 99.1% density,
- 63.1 vol.% packing sinters to 98.2% density,
- 61.5 vol.% packing sinters to 94.6% density.
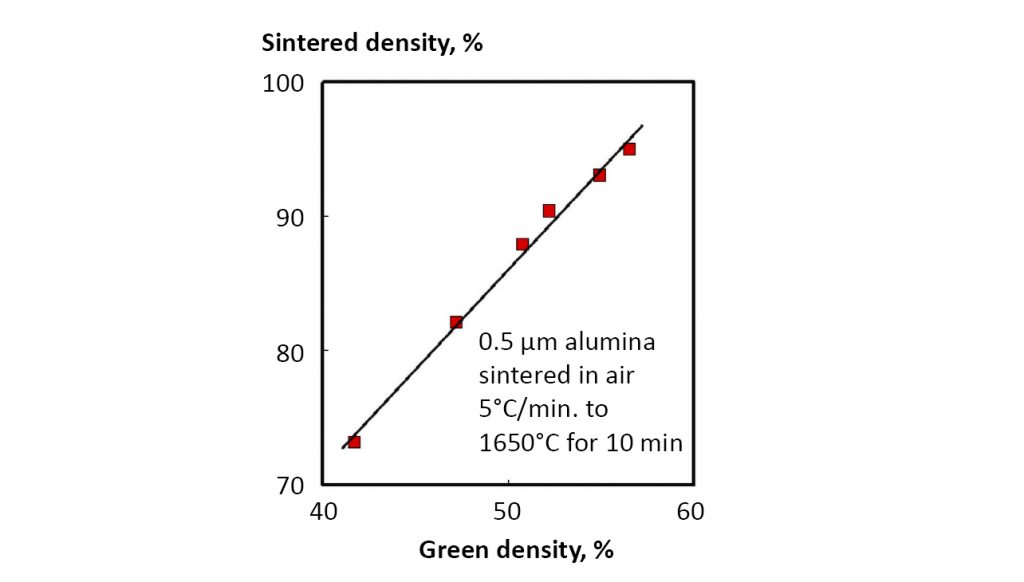
A high packing density helps sintering as illustrated in Fig. 13. The sintered density is plotted versus the green density for alumina powder subjected to the same sintering cycle. From a practical standpoint, if the green density falls below about 45%, then it is very difficult to reach full density.
Microstructure trajectory
Several microstructure parameters change during sintering. Besides neck growth and surface area loss, pores round and coalesce while grains grow. Grain growth involves atomic exchange across grain boundaries and accelerates on approaching full density. Pores slow growth since they reduce the grain boundary area. Hence, the slowest rate of grain growth is during initial heating, when there is little grain-grain contact. As necks emerge and pores round, the higher grain boundary area allows for more grain growth.
During isothermal sintering, grain growth occurs with the median grain volume (G3) increasing as follows:
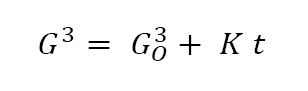
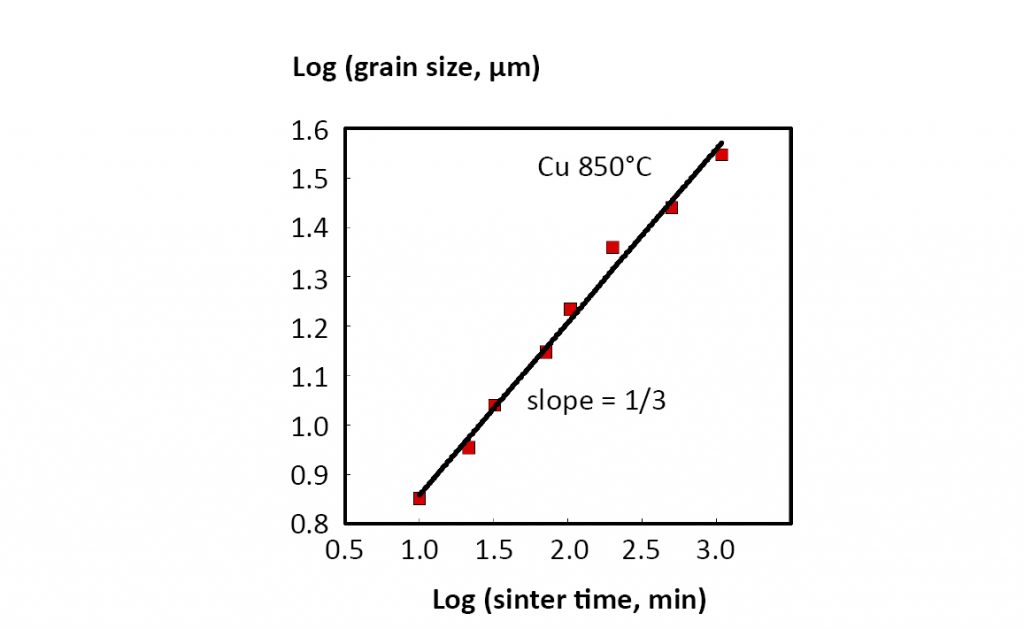
This says the median grain volume depends on the initial grain volume GO3, hold time t, and a temperature dependent parameter K. As a demonstration of this behaviour, 850°C sintering data for copper are plotted on a log-log basis in Fig. 14. The slope of one-third corresponds to G3 varying with t. Grain size is important to several properties.
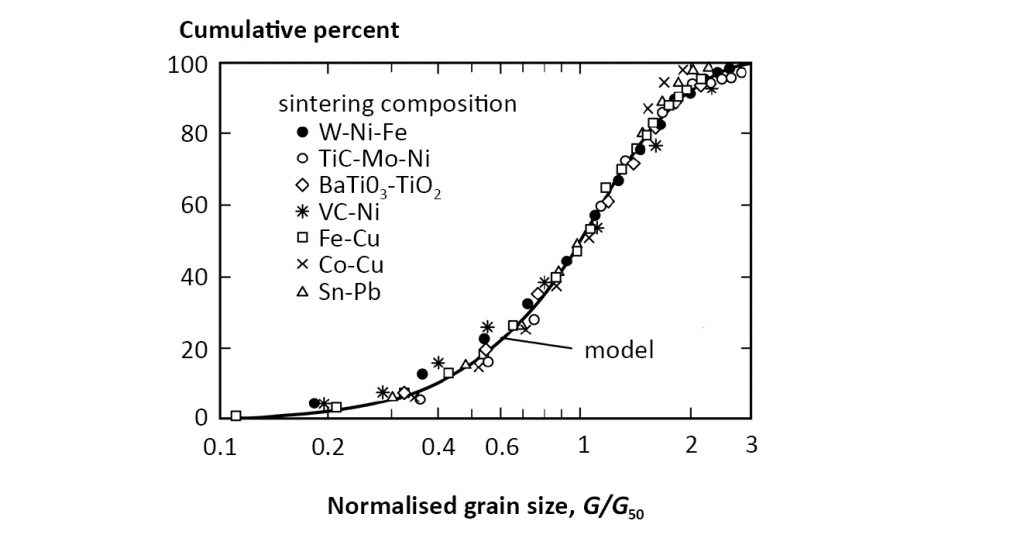
The sintered grain size distribution is self-similar, meaning the distribution shape is the same no matter the median grain size. Example sintered gain size distributions are plotted in Fig. 15 for a wide range of materials. The size scale is normalised to the median grain size. The symbols are experimental measures from a variety of sintered materials, while the solid line corresponds to a fit; F(G) is the cumulative fraction of grains with size of G, where G50 is the median size (half the grains are larger and half are smaller),
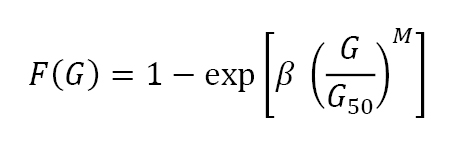
This is a Weibull distribution with β = -ln2 (or -0.6931), ensuring F(G) = 0.5 at the median grain size when G = G50. The exponent M reflects the distribution dispersion and is usually near 3.
Initially, grain growth is slowed by pores since vapour transport is relatively slow (evaporation and condensation) so grain boundaries encounter a drag force from the pores. As a result, a relation emerges between sintered median grain size G50 and fractional density f,
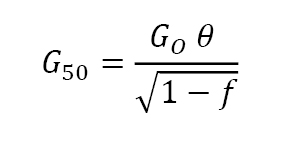
Again GO is the initial grain size (effectively the particle size) and θ is typically near 0.6. This relation is invalid when the sintered material reaches full density because grain growth continues without a change in porosity. Fig. 16 plots data from 4.3 µm nickel powder sintered at 900°C to reach a final density of 93% to show agreement with this equation.
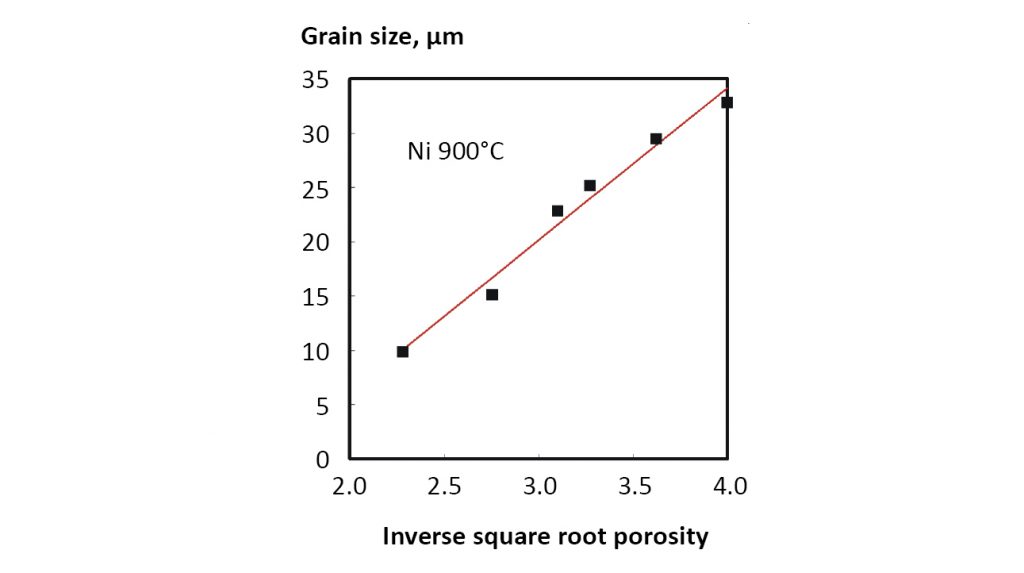
Because sintering results in a self-similar microstructure we find the pore size d is a function of fractional porosity ε and grain size G as follows:
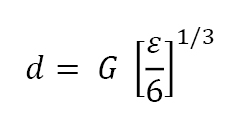
Grain growth leads to a pore size increase while porosity and the number of pores decline.
Pores and grains are coupled to one another during sintering; thus, the pore size also conforms to a Weibull distribution. The cumulative fraction of pores F(d) larger than size d depends on the median pore size d50 and an exponent M. The corresponding distribution is expressed as,
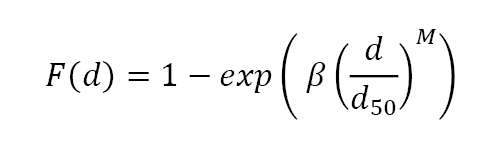
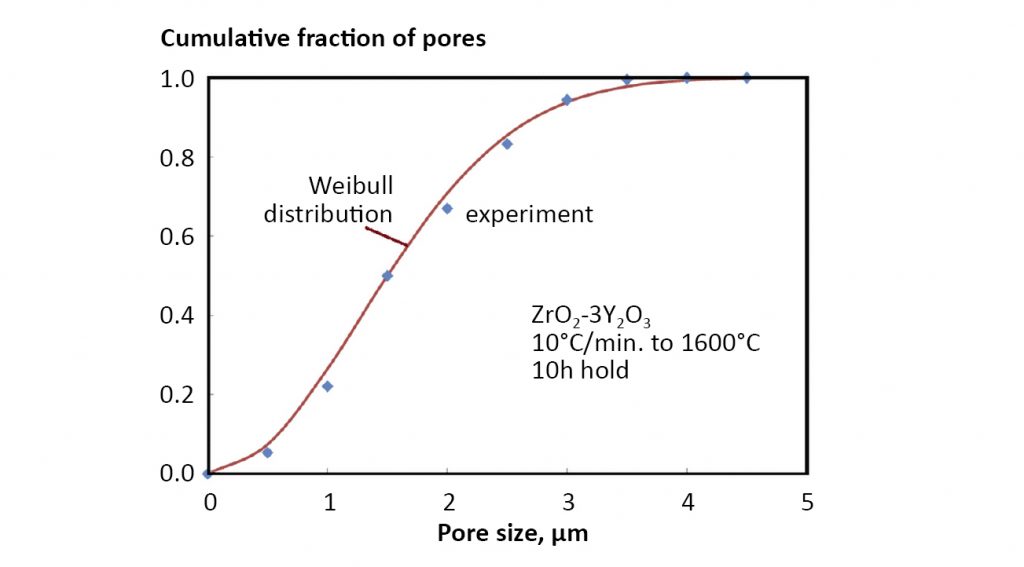
Again β equals -0.6931. Fig. 17 plots cumulative pore size distribution data for sintered zirconia according to this relation. The solid line is the fit to the pore size data using M = 2. The coupling of pores to grain boundaries causes the pore size distribution to follow the same trajectory as the grain size distribution.
Thus, during sintering the microstructure evolves, where the median grain size enlarges, the number of grains decreases, porosity declines, but pores coalesce and enlarge. The microstructure is self-similar, meaning the distributions in grain size, grain shape, and pore size generally track the same characteristics independent of the time-temperature, material, and processing factors.
Effects of key sintering parameters
Most applications for sintered metals are structural components requiring attention to mechanical properties. Sintered surgical stainless steel illustrates the key parameters and their impact on properties. The common alloy is 17-4 PH, formally designated AISI 630. It is a precipitation hardened martensitic stainless steel that gives a good combination of strength and corrosion resistance [16-19].
Pre-alloyed 17-4 PH powders are available from 0.2 to 100 µm sizes, with 10 to 20 µm being most popular. Usually the particles are rounded or spherical to enable good packing to about 60% green density. For a typical 15 µm powder, a hold near 600°C during heating ensures binder removal. Sintering shrinkage starts about 900°C, so another hold near that temperature is appropriate to remove oxides while pores are open for evaporation. Hydrogen is able to penetrate pores from the surface, forming steam that egresses out of the body. Long hold times at the peak sintering temperature are not productive, since most densification occurs during heating. For example, at 1320°C the peak density is achieved within 10 min. Higher sintering temperatures induce grain growth and evaporative loss of chromium and copper, compromising corrosion resistance and strength. However, heat transfer in the sintering furnace takes several minutes, so a hold is needed to obtain uniform shrinkage throughout the furnace load. The high thermal conductivity of hydrogen helps ensure uniform heating.
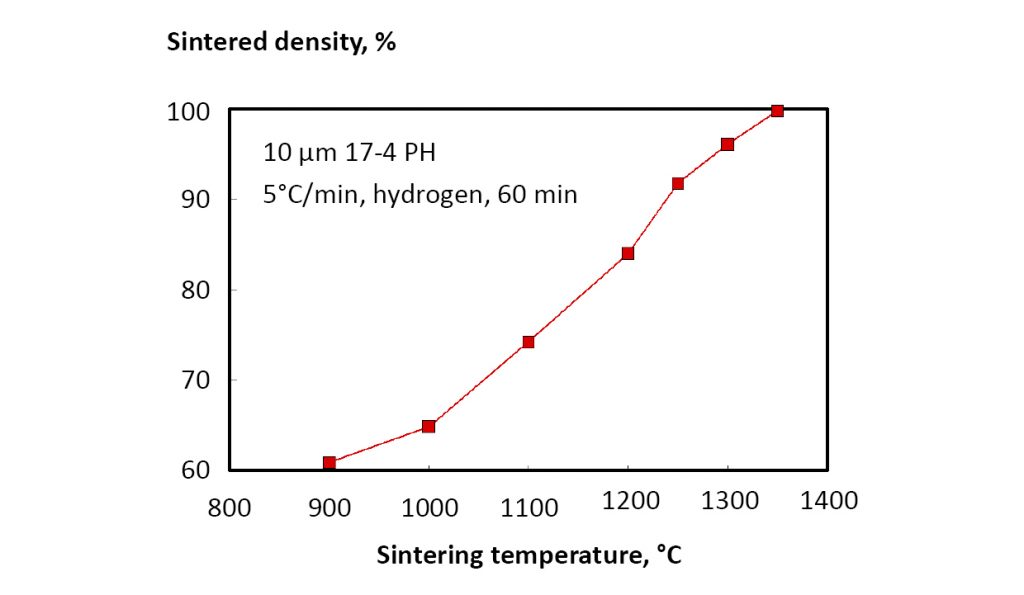
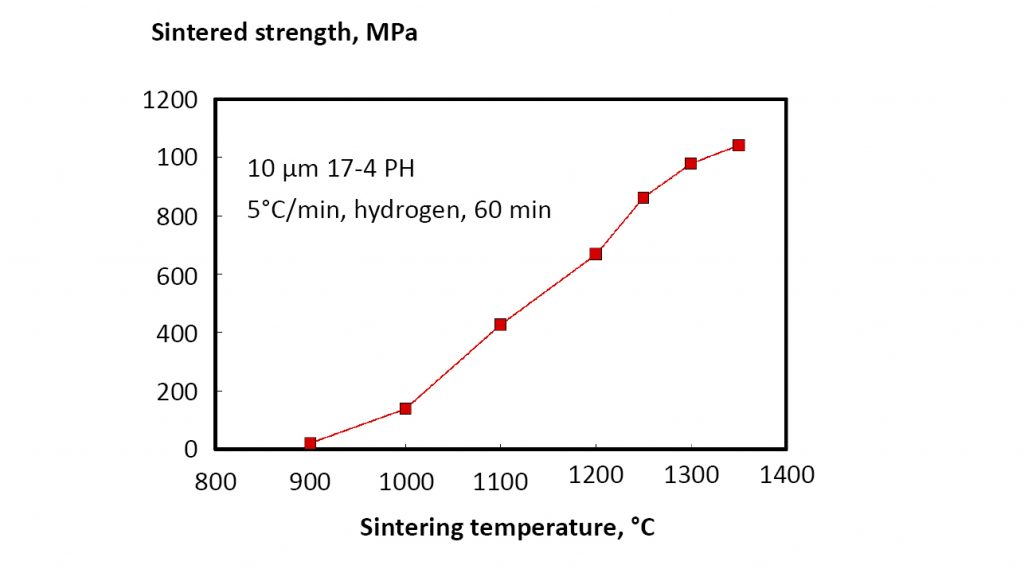
Strength increases with sintered density, so understanding the conditions for reaching at least 95% density is a key concern. Typical data for sintered density versus temperature are shown in Fig. 18 for a 10 µm powder (heated at 5°C/min. to temperature with 60 min. soak) [18]. In turn, sintered strength increases, as shown in Fig. 19. These results rely on hydrogen sintering; however, production atmospheres vary between operations and include vacuum, hydrogen, partial pressure hydrogen, argon, partial pressure argon, argon-hydrogen, and nitrogen-hydrogen. Argon is insoluble, so it inhibits densification once pores close, resulting in lower sintered density and properties. Nitrogen forms chromium nitrides, resulting in loss of corrosion resistance. Thus, high mechanical properties come from hydrogen or vacuum sintering, giving 99% density and heat treated hardness of 42 HRC, with 1317 MPa tensile strength and 10% elongation (1172 MPa yield strength) [16].
Graphite vacuum furnaces are successful for sintering 17-4 PH. Graphite reacts with residual oxygen to create a low partial pressure of carbon monoxide. Chromium oxide is reduced by carbon monoxide when the CO partial pressure is maintained at a low level, below about 100 ppm at temperatures over 1100°C. A similar principle operates in argon sintering in a graphite furnace, but the argon remains in the pores to inhibit final densification. For this reason, a low partial pressure of argon is an option to suppress evaporation with only a minor impediment to final densification [4].
As with other ferrous alloys, additives are effective in reducing the sintering temperature. Boron added as nickel boride allows sintering to full density at 1260–1280°C. After heat treatment to 52 HRC the tensile strength is 1400 MPa with 7% fracture elongation [17].
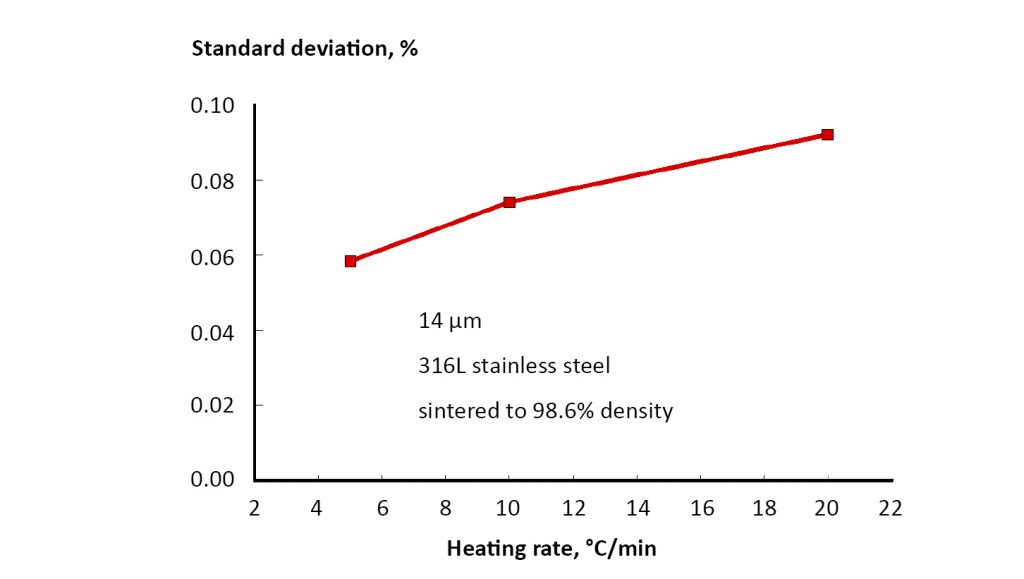
Sintered dimension uniformity is a concern, especially with large shrinkages. Dimensional variation arises from: anisotropic pore shapes in the green body, powder segregation in the forming process, thermal gradients in the furnace, gravity (especially for larger components), sticking or friction with the substrate, and mass variations between components. As an example, consider rapid heating. Heat transfer leads to temperature gradients in the furnace and in the component. The furnace centre is cooler than the edge, resulting in gradients that induce different dimensions depending on position in the furnace. Further, dimensional variation increased with heating rate as illustrated in Fig. 20 [14]. These data are for a 100 mm starting dimension formed using 14 µm 316L powder, sintered at 1385°C. Likewise, component mass variation, as often occurs in the forming step, is a precursor to differences in sintered dimensions between components [2,18].
Dimensional uniformity requires uniform green bodies and repeatable sintering cycles. Slow heating, uniform temperature, and sufficient hold time to thoroughly reach temperature are key to reduced dimensional variation. However, those measures cannot correct for variations introduced in the forming step.
This article hints at the underlying science explanation for sintering. At the atom scale the atomic motion events are random, but the slight bias provided by interface energy results in a progressive elimination of first surface area and later grain boundary area. Component densification is a by-product of this energy reduction. At the component level, shrinkage occurs. Non-uniform shrinkage and dimensional variation is the by-product of nonuniform green body and gradients introduced during sintering. Improved dimensional control requires attention to several factors besides the time-temperature cycle. Indeed, sintering is often just the messenger about variations occurring prior to sintering.
Conclusions
Sintering is a thermal process applied to solid particles to bond, strengthen, and possibly densify the particles. The events responsible for sintering take place at the atomic scale. Some liquid is often formed during a sintering cycle, but liquid phase contents are kept to a minimum, under 15 vol.%. Thus, sintering is different from melting such as occurs with Electron Beam or Laser Powder Bed Fusion-based Additive Manufacturing. Liquid phase sintering is similar to brazing the particles together, where the solidified liquid glues the solid grains together into a coherent and strong composite.
Sintering occurs at high temperatures where atomic motion is active and the material is soft. At high temperatures, surface energies are sufficient to induce bonding by surface diffusion. Subsequently, densification occurs by grain boundary diffusion. A high green density increases the number of bonds per particle to increase the attractive force for better sintering. Smaller particles have a higher curvature that determines the capillary force known as the sintering stress. The sintering stress acts to pull the particles together. With small particles the diffusion distances are shorter, so they naturally sinter faster. Temperature is an important control parameter, and for most materials, active sintering is observed close to the melting range. Long hold times at the sintering temperature are not useful and extended holds result in over-sintering. While densification ends when porosity is eliminated, grain growth continues and even accelerates as porosity is eliminated. Thus, peak sintered properties are often found at less than full density while grain size is small. Note that additives are common in sintered materials, to either impede grain growth or improve densification.
Pore behaviour is an anomalous part of sintering. As porosity declines, the average pore size increases, but the number of pores decreases. Both pore size and grain size take on self-similar Weibull distributions. The role of atmosphere in sintering is involved; active atmospheres accelerate sintering and help attain high final densities. On the other hand, inert gases become trapped in the pores to retard final stage densification. Often, vacuum sintering with a partial pressure (hydrogen or argon) provides a good balance between the several factors of heat transfer, suppression of evaporation and oxide reduction.
Structural components dominate the applications for sintered metals and electronic components dominate the applications for sintered ceramics. Both mechanical and electrical properties are quite sensitive to residual porosity, so high sintered densities are mandatory. Dimensional control, reflecting an ability to replicate final size with a small variation, is a major challenge in sintering. Although dimensional scatter is often assigned to the sintering step, in reality the source of scatter arises from poor control of the incoming powder or loose control of the shaping step – effectively sintering is the messenger, but not the cause.
Author
Professor Rand German is the most highly-cited researcher in sintering. He consults with firms in both Additive Manufacturing and Powder Injection Moulding, and serves as consulting editor for Powder Injection Moulding International. At San Diego State University, his current experiments are literally out of this world, involving microgravity liquid phase sintering on the International Space Station.
References
[1] A. S. Helle, K. E. Easterling, M. F. Ashby, “Hot Isostatic Pressing Diagrams: New Developments,” Acta Metallurgica, 1985, vol. 33, pp. 2163-2174.
[2] R. M. German, Sintering: From Empirical Observations to Scientific Principles, Elsevier, Oxford, UK, 2014.
[3] R. F. Wang, Y. X. Wu, X. Zhou, C. A. Tang, “Debinding and Sintering Processes for Injection Molded Pure Titanium,” Powder Metallurgy Technology, 2006, vol. 24, no. 2, pp. 83-93.
[4] A. Bose, I. Otsuka, T. Yoshida, H. Toyoshima, “Metal Injection Molding of Ultrafine 316L Stainless Steel Powders,” Advances in Powder Metallurgy and Particulate Materials – 2007, Metal Powder Industries Federation, Princeton, NJ, 2007, pp. 4.57-4.68.
[5] R. M. German, Metal Injection Molding – A Comprehensive MIM Design Guide, Metal Powder Industries Federation, Princeton, NJ, 2011.
[6] H. I. Bakan, D. Heaney, R. M. German, “Effect of Nickel Boride and Boron Additions on Sintering Characteristics of Injection Moulded 316L Powder Using Water Soluble Binder System,” Powder Metallurgy, 2001, vol. 44, pp. 235-242.
[7] R. Tandon, R. M. German, “Sintering and Mechanical Properties of a Boron-Doped Austenitic Stainless Steel,” International Journal of Powder Metallurgy, 1998, vol. 34, no. 1, pp. 40-49.
[8] C. Tojal, T. Gomez-Acebo, F. Castro, “Development of PM Stainless Steels with Improved Properties through Liquid Phase Sintering,” Materials Science Forum, 2007, vol. 534, pp. 661-664.
[9] J. Takekawa, “Effect of Additions of B and Al2O3 Fine Powders on the Densification of SUS316L Steel Powder,” Journal of the Japan Society of Powder and Powder Metallurgy, 2003, vol. 50, pp. 807-811.
[10] H. O. Gulsoy, O. Er, M. L. Berkem, “Injection Molded of High Density 316L Stainless Steel for Bio Applications,” Advances in Powder Metallurgy and Particulate Materials – 2011, Metal Powder Industries Federation, Princeton, NJ, 2011, pp. 04.104-04.112.
[11] Standard Specification for Powder Metallurgy (PM) Boron Stainless Steel Structural Components, Standard B853, ASTM International, West Conshohocken, PA, 2016.
[12] R. M. German, “Computer Simulation of the Sintering Densification of Prealloyed Tool Steel Powders,” Journal of Materials Synthesis and Processing, 1997, vol. 5, pp. 379-390.
[13] R. Watanabe, Y. Masuda, “Quantitative Estimation of Structural Change in Carbonyl Iron Powder Compacts during Sintering,” Transactions of the Japan Institute of Metals, 1972, vol. 13, pp. 134-139.
[14] G. J. Shu, K. S. Hwang, Y. T. Pan, “Improvements in Sintered Density and Dimensional Stability of Powder Injection Molded 316L Compacts by Adjusting the Alloying Composition,” Acta Materialia, 2006, vol. 54, pp. 1335-1342.
[15] E. A. Barringer, H. K. Bowen, “Formation, Packing, and Sintering of Monodisperse TiO2 Powders,” Journal of the American Ceramic Society, 1982, vol. 65, pp. C199-C201.
[16] M. Shirooyeh, “High-Performance 3D Printed Stainless Steel: A Metallurgical Perpspective on 3DEO’s Intelligent Layering,” Advances in Powder Metallurgy and Particulate Materials – 2019, Metal Powder Industries Federation, Princeton, NJ, 2019, on CD.
[17] H. O. Gulsoy, S. Salman, “Microstructure and Mechanical Properties of Injection Molded 17-4 PH Stainless Steel Powder with Nickel Boride Additions,” Journal of Materials Science, 2005, vol. 40, pp. 3415-3421.
[18] H. J. Sung, T. K. Ha, S. Ahn, Y. W. Chang, “Powder Injection Molding of a 17-4 PH Stainless Steel and the Effect of Sintering Temperature on its Microstructure and Mechanical Properties,” Journal of Materials Processing Technology, 2002, vol. 130, pp. 321-327.
[19] B. Julien, M. Ste-Marie, R. Pelletier, F. Lapointe, S. Turenne, “Parametric Modeling of MIM As-Sintered Properties of Stainless Steel 17-4 PH and 316L Using a Statistical Model,” Advances in Powder Metallurgy and Particulate Materials – 2010, Metal Powder Industries Federation, Princeton, NJ, 2010, pp. 4.1-4.10.




















