Health Canada approves first Canadian AM medical implant
December 7, 2021
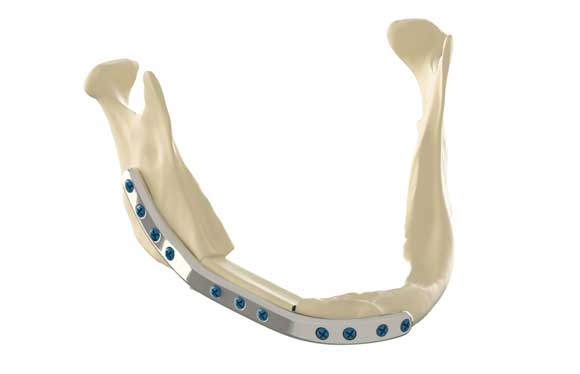
Health Canada has approved its first Canadian additively manufactured medical implant, created at the 3D Anatomical Construction Laboratory (LARA 3D) at Investissement Québec – CRIQ’s facilities with support from university hospital CHU de Québec-Université Laval. The bespoke 3D Specifit mandibular (lower jaw) plate is manufactured with biocompatible metals, specifically titanium grade 23 (Ti6Al4V-ELI), using Laser Beam Powder Bed Fusion (PBF-LB) for the implant and Electron Beam Powder Bed Fusion (PBF-EB) for the two surgical guides (cutting and drilling). It will be used for reconstruction in patients with oral cancer.
Launched in late 2020, LARA 3D has been ISO 13485 certified since April of this year. With Health Canada’s approval, issued in September, the green light has been given to treat patients and produce the 3D Specifit mandibular plate along with surgical cutting and drilling guides. This upgrade to patient-specific prostheses which are produced via AM rather than traditional manufacturing is anticipated to improve the quality of life for patients by reducing both surgery and recovery times.
“We are extremely proud to announce Health Canada’s approval of the Specifit 3D mandibular plate,” stated Dr Gaston Bernier D.M.D., Medical chief, Dental Medicine & Oncology, CHU de Québec-Université Laval. “Not only will it improve patients’ quality of life; but thanks to optimised, guided, and personalised surgery; it will also enable the development of a 3D medical equipment centre of expertise at Centre de recherche du CHU de Québec – Université Laval. We are convinced the approval of this technology marks only the start of innovation, research, and development in 3D medical printing at LARA 3D.”
As well as the work performed by LARA 3D, the orthopaedic screw manufacturer Alkom Digital and the producer of metal powders for AM AP&C, a GE Additive company, were also involved.
Lyne Dubois, vice-president, Investissement Québec – CRIQ, added, “With LARA 3D, Investissement Québec – CRIQ fully embraces its leadership role in Additive Manufacturing in Québec. The Specifit line of 3D prostheses will meet the needs of the healthcare sector and provide improved quality of life for patients. It also demonstrates the wide range of possibilities 3D printing affords companies that must design complex, light, and durable parts. The expertise acquired by Investissement Québec – CRIQ is accessible to the entire 3D-printing ecosystem in Québec. This will spur the development of innovative products and foster the growth of innovation companies.”
















