Paragon 28 acquires assets of Additive Orthopaedics
June 1, 2021
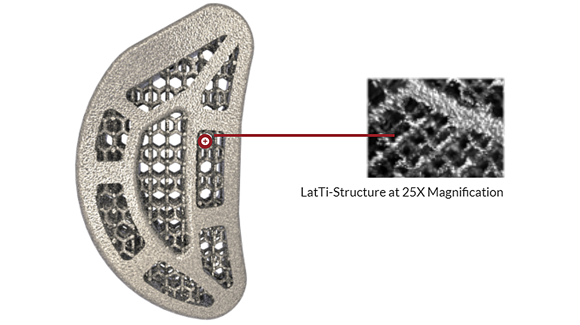
Paragon 28, Inc, Englewood, Colorado, USA, a market-leading orthopaedic medical device company focused exclusively on the foot and ankle, has announced that it has acquired the product lines of Additive Orthopaedics.
Additive Orthopaedics’ additively manufactured, patient-specific talus spacer was approved on February 17, 2021, by the US Food and Drug Administration (FDA) as a humanitarian use device. It is reportedly the first and only patient-specific total talus replacement implant authorised for use in the US. The implant is designed to replace the talus, the bone in the ankle that connects the leg and foot, and provides patients access to a novel, joint-sparing alternative to traditional ankle fusion therapies.
Also acquired as part of the transaction was Additive Orthopaedics’ internally developed, proprietary pre-operative surgical planning application. The application is an end-to-end, fully integrated cloud-based communication tool that aligns surgeons and engineers to design patient-specific surgical plans and implants, optimising patient outcomes.
“The addition of the Additive Orthopaedics product portfolio and surgical planning capabilities provides Paragon 28 customers exclusive access to the only FDA-approved patient specific total talus replacement implant,” stated Albert DaCosta, co-founder and Chief Executive Officer of Paragon 28. “The Additive acquisition also significantly accelerates the company’s strategy to leverage smart tools, artificial intelligence and advanced technology to improve patient outcomes.”
















