Materialise receives first US clearance for metal AM maxillofacial implants
September 21, 2017
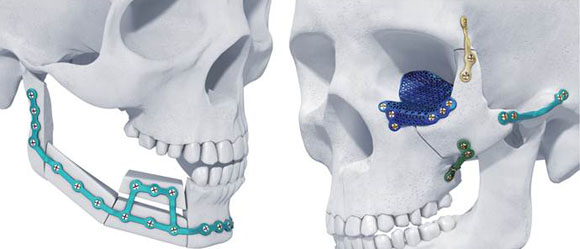
Materialise’s TRUMATCH titanium AM maxillofacial implants (Courtesy Materialise)
Materialise, Leuven, Belgium, has received US clearance for its additively manufactured titanium maxillofacial implants. The surgical implants will be distributed to the US market by DePuy Synthes, Westchester, Pennsylvania, USA.
TRUMATCH® titanium AM implants enable surgeons to provide patient-personalised solutions for orthognathic surgery, also known as corrective jaw surgery, as well as for facial reconstruction. They are produced using Materialise’s design software, clinical engineering and metal Additive Manufacturing production facilities, and, according to Materialise, are the first AM titanium maxillofacial implants to receive clearance for the US market.
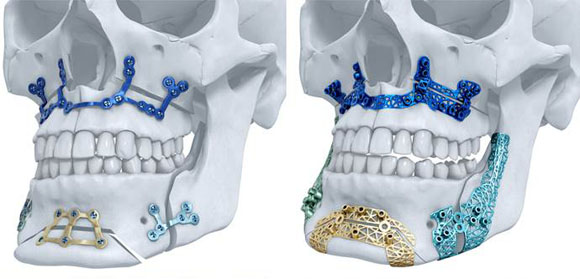
Materialise’s TRUMATCH titanium A maxillofacial implants (Courtesy Materialise)
By enabling the production of patient-specific virtual surgical planning, AM surgical guides and implants, Materialise’s system is said to help achieve better aesthetic results and minimise surgery time for patients. TRUMATCH has reported positive results in European and Australian markets since its introduction in 2016.
“As the first of our extensive selection of implants to receive clearance for the US markets, the decision is a real milestone for our medical department,” stated Brigitte de Vet, Vice President of Materialise Medical. “Thanks to our partnership with DePuy Synthes, our devices will be able to provide better healthcare for as many patients as possible.”
Dr Thomas Schouman, who uses TRUMATCH maxillofacial implants as CMF surgeon at Groupe Hospitalier Pitié Salpêtrière, France, commented, “For seven years now I’ve experienced the benefits of 3D printed implants first hand – they simplify maxillofacial surgery and allow me to perform procedures more accurately, saving time in the OR and improving patient outcomes.”
“Moreover, they offer new treatment possibilities, allowing me to perform more complex surgeries or multiple procedures in a single intervention whereas without the implants several interventions would be necessary.”
















