Exactech transitions shoulder implant manufacture from electron beam to laser beam AM
July 21, 2020

Orthopaedic device developer Exactech, Gainesville, Florida, USA, has announced that it is transitioning the production of its Equinoxe® Stemless Shoulder orthopaedic implant from Electron Beam Powder Bed Fusion (PBF-EB) to Laser Beam Powder Bed Fusion (PBF-LB) metal Additive Manufacturing.
The Stemless Shoulder was developed using PBF-EB and launched in 2018, after receiving 510(k) clearance from the US Food and Drug Administration. The company stated that, while it was able to produce successful implants using PBF-EB and has received positive clinical feedback, the transition to PBF-LB will enable it to scale up and increase its production volumes.
Designed for anatomic total shoulder arthroplasty, the implant system comprises a single tray of instruments for operating room efficiency and uses a three-dimensional porous structure said to offer optimised pore size, count and porosity for osseointegration (bone in-growth). In addition, the Stemless Shoulder incorporates a bone cage for bone through-growth to increase the likelihood of biologic fixation.
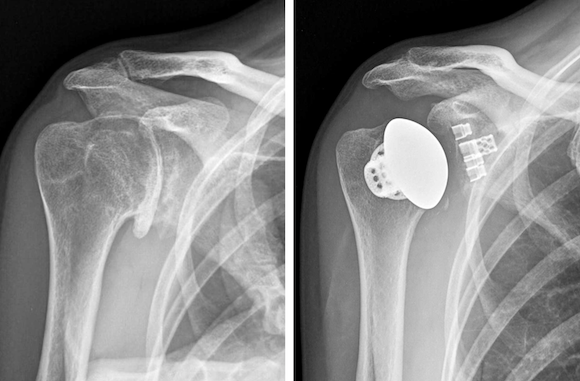
The first surgeries to implant the PBF-LB Stemless Shoulder were conducted early in July 2020 by orthopaedic surgeons Dr Curtis Noel, of the Crystal Clinic in Akron, Ohio, USA, a member of the implant’s original design team, and Dr Stephanie Muh, of the Henry Ford Health System in Detroit, Michigan, USA.
“We have been incredibly pleased with our original EBM Stemless Shoulder implant and the early positive clinical feedback we received from our surgeon customers,” stated Chris Roche, Exactech’s vice president of Extremities. “The new laser-printed device is built on this solid foundation while also giving us the ability to ramp up production to serve even more patients, which drives us and fulfils our mission.”
Over the remainder of 2020, Exactech expects to transition all planned Stemless Shoulder implant procedures in the USA to use devices produced by PBF-LB.
















